Packages
Gender DNA Blood Test 6+ Weeks
$200 | 4-7 Day Results*
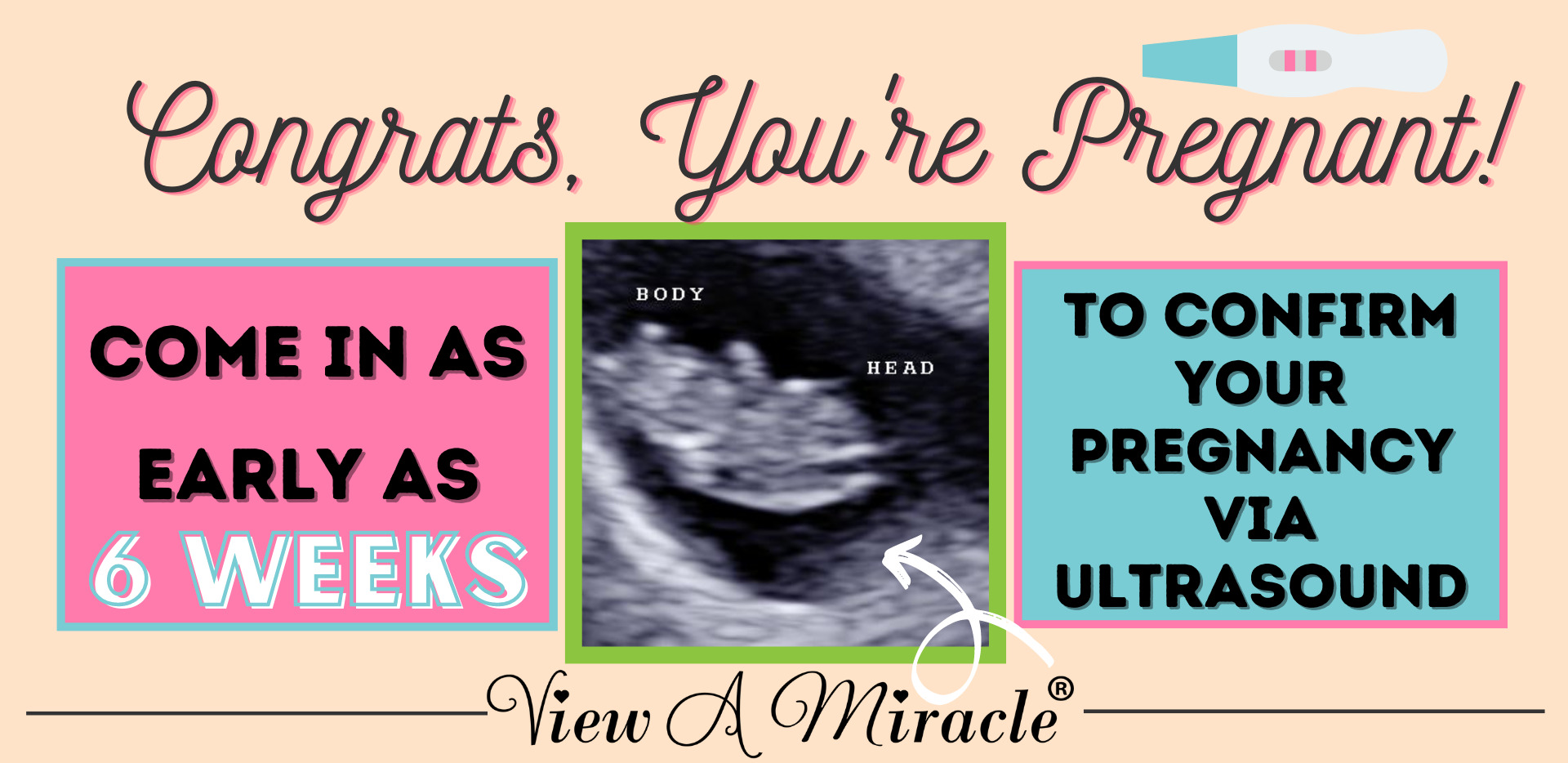
This package includes a transvaginal ultrasound to determine the baby is at least 6 weeks, to see the baby’s heart beat, as well as your blood drawn to send to the lab for Gender Determination. *The Gender Reveal results are emailed QUICKLY to you directly from the lab in just 4-7 business days. 2 business day fast results for fee of $50.